
The fundamental difference between the two methods is that molecular cloning involves replication of the DNA in a living microorganism, while PCR replicates DNA in an in vitro solution, free of living cells. You can then amplify the sequences using primers that bind to the adapter sequences. Molecular cloning is similar to polymerase chain reaction (PCR) in that it permits the replication of DNA sequence. (Examples where this can be used are when you have sequences from related organisms or amino acid but not nucleotide sequence.)Ī technique that can be used if you need to amplify restriction fragments is to ligate short "adapter" sequences onto the ends of the restriction fragments. If you don't know the exact sequence, but have some sequence information you can use "degenerate" primers, which are mixture of many similar (but non-identical) sequences. When the polymerase chain reaction (PCR) is used to amplify simultaneously multiple templates, preferential amplification of certain templates (PCR bias) leads to a distorted representation of. This is typically done based on knowing the sequence you are trying to amplify. PCR is usually used to amplify small quantities of DNA into a large enough amount to use for something else (e.g. Thus, there isn't enough "information" (sequence) present from knowing a restriction site to design a PCR primer. In contrast, primers are usually at least 18 nt long (often much longer) and so recognize a sequence that is on average at least 3 times longer. The recognition sequences for restriction enzymes are typically quite short (6 bp long is most usual). 
ligate it into a vector cut with the same REs). If you use restriction enzymes (REs) then you usually already have enough DNA and can gel purify and use the cut fragment directly (e.g. GCGGCG-restriction site-your sequence).You don't need to (and typically won't) cut the DNA before doing PCR. Note: If you will be including a restriction site at the 5’ end of your primer, note that a 3-6 base pair "clamp" should be added upstream in order for the enzyme to cleave efficiently (e.g. Primer pairs should not have complementary regions Primer pairs should have a Tm within 5☌ of each other.Taking into consideration the information above, primers should generally have the following properties: 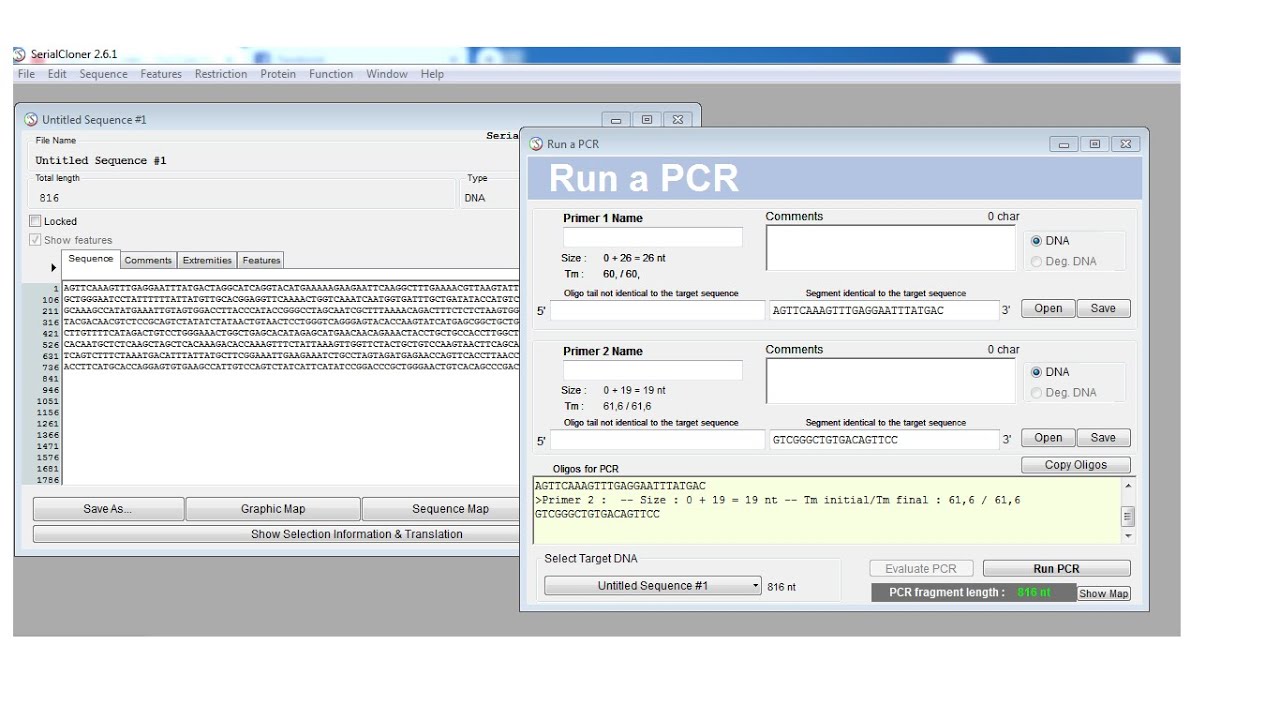
One can also use a nucleotide-based molecular insert (inosine) instead of a regular nucleotide for broader pairing capabilities. When designing, if unsure about what nucleotide to put at a certain position within the primer, one can include more than one nucleotide at that position termed a mixed site. One also needs to avoid primer-primer annealing which creates primer dimers and disrupts the amplification process. The structure of the primer should be relatively simple and contain no internal secondary structure to avoid internal folding. On average, the DNA fragment that needs to be amplified should be within 1-10 kB in size. Short primers produce inaccurate, nonspecific DNA amplification product, and long primers result in a slower hybridizing rate. However, a primer should not be too long (> 30-mer primers) or too short. On the other hand, a long primer is used to amplify a eukaryotic genomic DNA sample. Short primers are mainly used for amplifying a small, simple fragment of DNA. The size of the primer is very important as well. Also, both of the 3’ ends of the hybridized primers must point toward one another. Usually a guanine or cytosine is used at the 3’ end, and the 5’ end of the primer usually has stretches of several nucleotides. However, primers do not need to correspond to the template strand completely it is essential, however, that the 3’ end of the primer corresponds completely to the template DNA strand so elongation can proceed. The main property of primers is that they must correspond to sequences on the template molecule (must be complementary to template strand). One must selectively block and unblock repeatedly the reactive groups on a nucleotide when adding a nucleotide one at a time. They are synthesized chemically by joining nucleotides together. 
One needs to design primers that are complementary to the template region of DNA. Simply input the DNA sequences of your vector and insert (s), along with your linearization method to generate primers for your next cloning. Oligonucleotide primers are necessary when running a PCR reaction. Our NEW In-Fusion Cloning Primer Design Tool allows for single- or multiple-insert cloning, accommodates vector linearization by inverse PCR or restriction digest, and enables site-directed mutagenesis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
